
Barcelona, Spain, 20 February 2026 — Held at the Institut d’Estudis Catalans, the meeting was officially opened by Iban Ubarretxena, Scientific Coordinator of Euro-BioImaging Spain, together with Inmaculada Figueroa (Subdirector General for Scientific and Technical Infrastructures and Large Facilities, Ministry of Science, Innovation and Universities, Spain) and John Eriksson, Director General of Euro-BioImaging ERIC. Their opening remarks highlighted the strategic importance of imaging technologies for advancing scientific excellence and fostering innovation across Europe. Building on this vision, the scientific sessions brought together leading experts in both biological and medical imaging, showcasing cutting-edge developments across scales—from molecular and cellular imaging to mesoscopic and in vivo approaches, and from preclinical to clinical applications—highlighting the transformative role of imaging in addressing complex biological and biomedical challenges, including the advancement of more personalised approaches to healthcare.
Bringing Light to Life: Advanced in Optical Imaging and Photonics
The session continued with a keynote by James Sharpe (EMBL), who presented how multiscale quantitative imaging has become a driving force for dynamical computer modelling of organogenesis. Sharpe explained how his team combines mesoscopic imaging techniques—such as Optical Projection Tomography, which he originally pioneered—with computational modelling to capture the cellular behaviours that shape organ development. By producing volumetric time-lapse datasets that map millions of cells across large biological structures, his group is able to extract quantitative measurements of growth, deformation and patterning. These high-content datasets feed into predictive models capable of simulating organ formation under different mechanical and biochemical conditions. Sharpe emphasised that, while omics techniques provide valuable molecular information, they lack the crucial spatial context required to understand how tissues actually take form. Imaging, he argued, supplies the missing link by enabling large-scale, spatially resolved, dynamic measurements needed to build mechanistic models of development—models that can ultimately explain, and even predict, how complex biological shapes emerge.

Following this, María García-Parajo (ICFO – The Institute of Photonic Sciences) showcased how advanced nanoscale imaging approaches are revealing the spatial and temporal organisation of biomolecules in living cells with unprecedented resolution. She described how her group pushes the boundaries of super-resolution microscopy and single-molecule tracking to uncover nanoscale structures that remain invisible to traditional fluorescence imaging. Through techniques capable of achieving nanometre precision and millisecond temporal resolution, her team has identified dynamic molecular assemblies, membrane nanodomains and transient multi-molecular interactions critical for cell signalling. García-Parajo emphasised that fixed-cell imaging, while valuable, captures only a static snapshot and can obscure fast, transient biological events. To address this, her group developed live-cell single-particle tracking methodologies that enable real-time observation of processes such as viral trafficking, including how HIV navigates through dendritic cells. She also presented evidence showing how receptor nanoclustering enhances cellular recognition and signalling. Her work highlights how nanophotonics and molecular-scale imaging open new avenues to understand cell function at the level of individual molecules
AI Transforming Healthcare: Biomedical Imaging and Advanced Radiology
The programme then shifted toward clinical imaging, where artificial intelligence has been rapidly gaining ground
Luis Martí-Bonmatí (GIBI230 –Instituto de Investigaciones Sanitarias IIS La Fe) delivered a critical reflection on how medical imaging must evolve to ensure that AI-derived findings translate into clinically meaningful evidence. His presentation addressed what he termed the “broken telephone effect” in imaging workflows, where each step—from acquisition to analysis—introduces potential distortions that compromise reproducibility and reliability. Martí-Bonmatí emphasised that imaging biomarkers and AI-based predictions often rely on ground truth labels that are imperfect or inconsistent, and he warned about overconfidence in image-based pattern recognition that lacks causal understanding. He highlighted the variability introduced by different scanners, protocols, patient populations and study designs, arguing for the adoption of transparent analytical workflows, rigorous bias control and harmonisation strategies across centres. He also stressed the importance of independent validation and causal reasoning frameworks, explaining that clinical deployment requires not just accurate models but models that reflect true biological mechanisms. His talk positioned imaging AI as a powerful but fragile tool that must be rigorously managed to benefit patients safely and equitably.

Raquel Pérez-López (VHIO Vall d’Hebron Institut d’Oncologia)presented an in-depth overview of how artificial intelligence is reshaping oncologic imaging and accelerating the development of personalised cancer care. She described how AI-driven tools are now supporting the full cancer continuum—from early detection through screening programmes, to tumour segmentation, treatment monitoring and prediction of therapeutic response. Pérez-López illustrated how advanced radiomics and voxel-level analysis allow clinicians to extract subtle imaging biomarkers that inform tumour biology, aggressiveness and prognosis. She also discussed emerging multimodal approaches that integrate imaging with clinical, genomic and pathology data, as well as the rise of foundation models capable of learning from diverse datasets and performing multiple downstream tasks. Importantly, she highlighted that the number of regulatory-approved medical-imaging AI tools is increasing, signalling momentum toward clinical adoption. However, she stressed the need for robust validation, prospective multicentre studies, standardised evaluation metrics and transparent reporting to ensure that AI systems are trustworthy, equitable and clinically safe. Her talk outlined a future in which imaging AI complements clinical expertise to deliver more precise and effective cancer care.
Building Imaging Networks: National BioImaging Infrastructures:Organization/Activities, Digital Transformation, Innovation / Training / Access
A central theme of the meeting was how national imaging infrastructures organise their capabilities and expertise to better support scientific communities.
During her presentation, Luisa Cortes, coordinator of the Portuguese BioImaging Infrastructure (PPBI), highlighted the crucial role of national collaboration in building a coordinated network that provides open access to cutting-edge imaging technologies and expert support. She explained how PPBI’s inclusion in the Portuguese Roadmap of Research Infrastructures enabled the harmonisation of equipment and capabilities across the country, generating a measurable impact on the scientific community. Cortes emphasised the need for proper acknowledgement of imaging facilities in research publications, noting that such recognition is essential to demonstrate their contribution to scientific progress. Training also featured prominently in her talk: she outlined PPBI’s extensive programme of specialised courses, distributed training formats, and joint initiatives between nodes. Finally, she showcased the infrastructure’s growing international engagement through collaborations with Euro-BioImaging, the organisation of major scientific meetings such as SPAOM and the upcoming ELMI 2026 in Coimbra, as well as new outreach actions aimed at expanding PPBI’s reach, including to Portuguese-speaking African countries.

From France, Emmanuel Faure (France BioImaging) presented how coordinated national infrastructures can accelerate innovation in imaging and data management. He described the scale and diversity of the France BioImaging network, which spans 11 cities and more than 30 imaging facilities, offering a rich ecosystem of technologies and expertise. Faure focused particularly on the infrastructure’s transversal bioinformatics node, which connects research and development teams across the country to address shared computational challenges. He explained how, in the absence of new funding, France BioImaging developed community-driven solutions such as a national image-analysis federation. This initiative brought together analysts from multiple sites to share expertise, avoid duplication of effort and jointly solve user problems. The “open desk” consultation programme, where experts dedicate one hour a month to help biologists tackle specific analysis questions, has become a successful model of distributed collaboration. Faure also highlighted efforts to implement FAIR-aligned data-management pipelines that link imaging instruments directly to national data centres, reducing the burden on local facilities and enabling long-term, scalable data handling. His talk demonstrated how well-structured infrastructures can amplify national capabilities and foster innovation even under constrained resources.

In his contribution, Alvaro Sahún presented Madrid Advanced Microscopy Center (MAdMiC), a pioneering Spanish node that has recently joined Euro-BioImaging as part of the organisation’s latest expansion. MAdMiC brings together the Advanced Optical Microscopy and Cryo-Electron Microscopy facilities at the Centro Nacional de Biotecnología (CNB-CSIC), along with the Advanced Optical Microscopy and Preclinical Biomedicine facilities of the Centro de Biología Molecular Severo Ochoa (CBM-CSIC-UAM). Sahún described how this integrated node—co-led by Ana Oña Blanco and Requejo-Isidro at CNB, —provides access to more than 26 state-of-the-art imaging systems, supporting over 25 distinct microscopy techniques.
He highlighted that MAdMiC will focus on capturing dynamic biological processes across multiple scales, enabling researchers to visualise structures ranging from nanometre-sized molecular complexes to centimetre-scale samples in full 3D. This new node significantly broadens Euro-BioImaging’s Spanish technological portfolio and strengthens national capacity for advanced training, user support and methodological innovation. Importantly, Sahún noted that MAdMiC also enhances Spain’s geographical representation within Euro-BioImaging by establishing a major hub in Madrid, complementing existing nodes in Barcelona, Bilbao and Valencia. His presentation underscored how the rigorous scientific evaluation and high technical standards required for Euro-BioImaging membership affirm MAdMiC as a high-quality, internationally competitive research infrastructure now available to the European imaging community.
Bridging Scales and Modalities: Image Analysis and Intelligence for Multimodal Image Data
Artificial intelligence returned to the forefront in a dedicated session on multimodal and multiscale data integration.
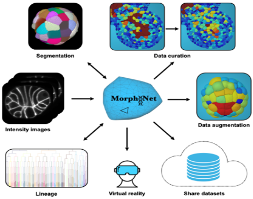
In this second contribution, Emmanuel Faure presented MorphoNet (https://morphonet.org/) , an integrated platform designed to address one of the central challenges in modern bioimaging: transforming increasingly complex 3D and 3D-time microscopy datasets into biologically reliable, curated information. Faure explained that while state-of-the-art segmentation algorithms such as Cellpose, Stardist or Ilastik can now generate large 3D reconstructions, the true bottleneck lies in validating, correcting and evaluating these outputs at scale. MorphoNet tackles this gap by offering a unified environment where users can visualise voluminous datasets, explore segmentation-derived surface meshes, compute geometric and intensity-based descriptors, and identify potential errors through an intuitive, interactive interface. Combining a Python-based processing backend with a Unity-powered 3D viewer, the platform enables seamless transitions between raw images, segmentations and curated structures.
A key innovation highlighted by Faure is MorphoNet’s approach to bio-curation, which moves beyond manual ground-truth annotation toward a modular, semi-automated workflow. Through its plugin system, users can apply targeted corrections—splitting, merging or reconstructing problematic regions—and even extend functionality through custom tools, fostering a community-driven ecosystem. He illustrated how these curated datasets, enriched with quantitative metrics such as volume, compactness or intensity heterogeneity, have improved biological interpretation in diverse models including zebrafish, Tribolium, starfish and Arabidopsis. Designed with FAIR principles in mind, MorphoNet also incorporates cloud-based sharing, collaborative visualisation and even virtual-reality exploration, enabling reproducible analyses across laboratories. Faure concluded that MorphoNet is more than a visualisation tool: it is a comprehensive framework that bridges automated reconstruction and expert validation, offering a scalable and accessible solution for managing the growing complexity of biological imaging data.

The session continued with a presentation by Ignacio Arganda-Carreras (IBF, Instituto Biofisika (CSIC-EHU), who introduced BiaPy, an open-source platform designed to make deep-learning-based bioimage analysis workflows accessible, reproducible and portable across computational environments. Arganda-Carreras explained how BiaPy addresses common barriers faced by biologists and developers alike—such as the difficulty of deploying state-of-the-art models, the need for reproducibility, and the challenge of adapting workflows to multi-dimensional microscopy data. The platform structures image-analysis workflows into modular pre-processing, deep-learning and post-processing components, enabling users to customise pipelines without requiring advanced programming skills. It supports a broad range of tasks, including 2D and 3D segmentation, object detection, image denoising, super-resolution, and even self-supervised learning for scenarios with limited annotations. Arganda-Carreras emphasised BiaPy’s multiple entry points—from a user-friendly GUI to notebook-based interfaces and API-level access—making it suitable for both novice users and expert developers. He also highlighted its integration with AI4Life’s BioImage Model Zoo, allowing researchers to directly import or share trained models. His contribution underscored the importance of creating accessible, community-driven tools that bring deep learning within reach for life-science researchers working with increasingly large and complex imaging datasets.

The session continued with a remote presentation by Leonor Cerdá-Alberich (Biomedical Imaging Research Group, La Fe Health Research Institute – Valencia), who discussed multimodal Artificial Intelligence (AI) modeling in oncology and the transformative concept of the “virtual biopsy”. She detailed how AI is increasingly being integrated into radiology to automate lesion detection and segmentation, extract tumor-related imaging biomarkers, and merge imaging with complementary data, such as clinical variables, genomics, and pathology, to enhance treatment-decision workflows.
Cerdá-Alberich showcased an end-to-end pipeline developed for pediatric cancer, where AI-based segmentation of neuroblastoma Magnetic Resonance (MR) imaging scans, combined with radiomics and computational growth simulations, enabled highly accurate prognostic modeling. She highlighted how these multimodal approaches outperformed conventional clinical risk groups in survival prediction, demonstrating AI’s potential to refine and personalize treatment strategies.Furthermore, she addressed in-silico validation frameworks for lung, breast, prostate, and colorectal cancers, alongside digital-twin models for prostate cancer and explainability techniques designed to build clinician trust. Her presentation concluded with future perspectives on foundation models and generative AI, emphasizing their capacity to integrate diverse data sources, mitigate dataset biases, and generate high-quality synthetic medical images. Ultimately, these advancements aim to provide radiologists and oncologists with trustworthy virtual assistants for more precise and ethical decision support.
Artificial Intelligence Across Scales: From Cells to Patients
The meeting concluded with a round table featuring María García-Parajo,Raquel Pérez-López, Aikaterini Vraka and Ignacio Arganda-Carreras, who discussed several of the key questions surrounding the use of artificial intelligence in imaging.

The conversation began with AI-based denoising: participants noted that modern algorithms can clean noisy images, recover weak signals and enable faster, lower-dose acquisitions, but at the cost of a potential risk of “hallucinations” or fabricated details. Rather than assuming hallucinations could be completely avoided, the panel argued for defining acceptable error rates, quantifying uncertainty and using appropriate metrics to assess whether artefacts actually impact the biological or clinical conclusions. They also stressed that expert interpretation, explainability methods and proper clinical validation remain essential safeguards, since AI adds a powerful layer of capability but also a new layer of responsibility.
The discussion then moved to data formats, standardisation and resolution enhancement. Using examples from medical imaging, where DICOM has long enabled interoperability, the panel contrasted this with the more fragmented landscape of biological imaging formats. They considered whether foundation models and advanced AI could eventually reduce the sensitivity to variations in protocols and formats, and even help extract value from legacy datasets such as old electron-microscopy negatives with poor or missing metadata.
Finally, the panel addressed AI-driven super-resolution and cross-modal image reconstruction, acknowledging the exciting possibility of “democratising” advanced microscopy by compensating for hardware limitations. At the same time, they warned that many current methods work well on simple, well-known structures but may be risky when applied to unknown or highly complex architectures. Overall, the round table highlighted both the enormous potential of AI to extend what imaging can do and the need for rigorous validation, careful interpretation and continued human expertise to ensure that these tools are used in a trustworthy and meaningful way.
A Community Moving Forward Together
The Euro-BioImaging Spain meeting demonstrated how rapidly the imaging landscape has evolved—and how crucial coordinated infrastructures, advanced technologies and AI-powered analysis have become for both research and healthcare. Across disciplines, speakers highlighted a collective goal: to make cutting-edge imaging tools accessible, interoperable and impactful for the scientific and clinical communities they serve.
Euro-BioImaging Spain, together with Euro-BioImaging ERIC and national partners, continues to work toward strengthening this ecosystem, promoting collaboration and ensuring that imaging remains central to Europe’s scientific and technological leadership.
The event was made possible with the support of AINOVIS, EVIDENT, NordicNeuroLab, Palex, Quibim, Rapp, Telstar and Zeiss, whose collaboration contributed to its successful organisation.
For more information
For further details about Euro-BioImaging Spain, its imaging services, access procedures or communication-related enquiries, please contact:
Alejandra Pinedo
Communication Office – Euro-BioImaging Spain
📞 +34 676 751 102
✉️ info@eurobioimaging-spain.es
